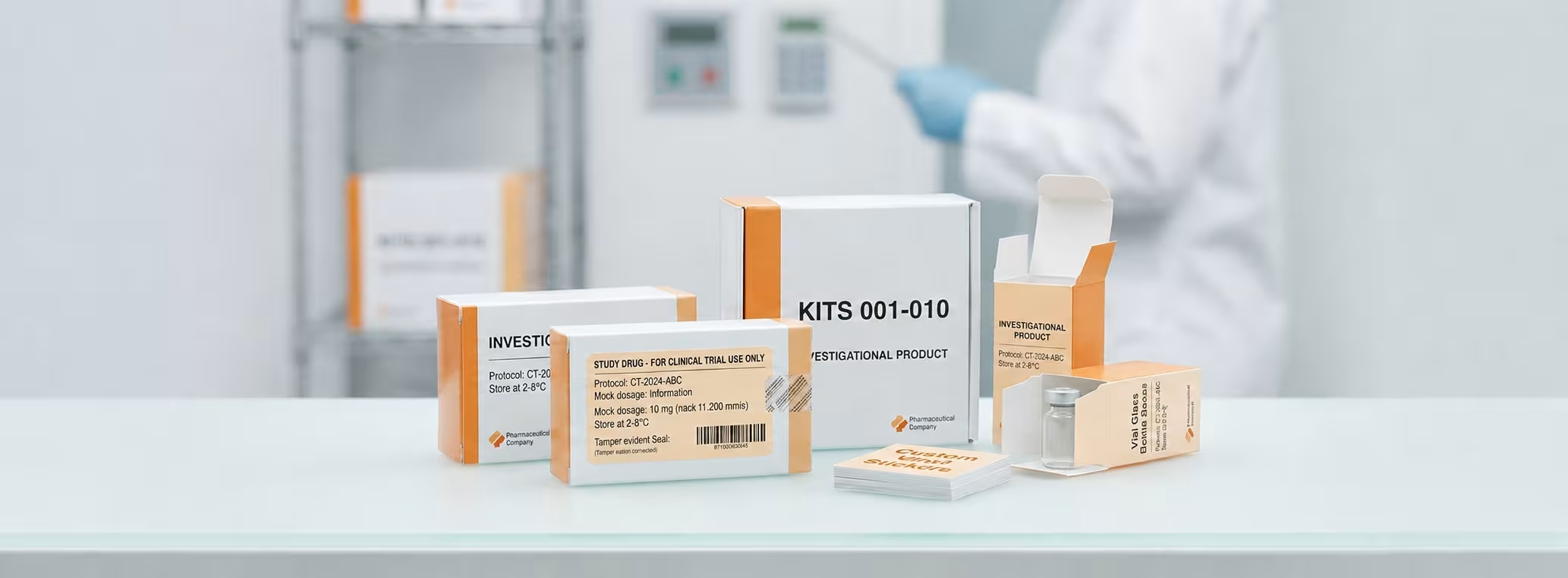
In the United States, clinical trials operate under strict systems designed to protect patients and produce reliable results. The packaging used for investigational drugs is a critical part of that system. It is not only used to contain the medicine, but also to support safety, accuracy, and legal compliance.
The packaging of investigational drugs should ideally be designed in a way that removes confusion, protects the drug from damage, and ensures that every participant receives the correct treatment. When packaging is done properly, it helps researchers maintain control over the study and avoid costly errors.
For companies like Sire Printing, understanding these expectations is important when providing packaging solutions for the pharmaceutical and healthcare industries in the U.S.
Strict Packaging Standards for Investigational Drugs in the U.S.
Investigational drugs are still under testing and are not approved for public use. Because of this, they must follow strict investigational drug packaging requirements that focus on safety and control.
In the U.S., these standards are regulated by the U.S. Food and Drug Administration. The agency requires that every part of the packaging process supports patient safety and ensures that no errors occur during the trial.
If packaging fails to meet these standards, it can lead to serious problems. Trials may be delayed, data may become unreliable, and in some cases, patient safety may be at risk. This is why packaging is treated as a core part of the clinical process, not just a supporting element.
How Packaging Supports Accurate Clinical Trial Results
-
Importance of Accuracy in Clinical Trials
Accuracy is the foundation of every clinical trial. The packaging must help ensure that each patient receives the correct medication without confusion.
- Role of Clinical Trial Drug Packaging Guidelines
This is where clinical trial drug packaging guidelines play an important role. These guidelines help maintain consistency across all trial locations and prevent mix-ups between different treatments.
- Blinded Study Requirements and Packaging Consistency
In many studies, patients and researchers do not know which treatment is being given. This is known as a blinded study. In such cases, packaging must look identical for all versions of the drug. This avoids bias and keeps the results fair and scientifically valid.
- Tracking and Control Across the Supply Chain
Proper packaging also helps track each unit from production to final use. This level of control is essential for maintaining trust in the trial results.
The Role of Labeling in U.S. Compliance
Labeling is one of the most sensitive parts of investigational medicinal product packaging. Every label must provide clear and accurate information without overwhelming the user.
In the United States, labeling must follow strict pharmaceutical labeling compliance USA standards. These rules ensure that patients and healthcare providers can understand how to use the product safely.
Label Requirements for Investigational Drugs
-
The label usually includes key details such as dosage instructions
-
It also includes storage conditions to ensure product stability
-
A clear statement is added that the product is for investigational use only
- If any of this information is missing or unclear, it can lead to misuse or regulatory issues
Ensuring Durable and Clear Label Quality
-
To maintain label quality, many companies use Custom Vinyl Stickers
-
Custom Vinyl Stickers are durable and remain readable under different storage conditions
- They help ensure compliance and reduce labeling errors in clinical trials
Protecting Drug Stability During the Trial
Investigational drugs can be sensitive to environmental conditions. Exposure to heat, light, or moisture can reduce their effectiveness. Because of this, packaging must provide strong protection at every stage.
The FDA clinical supply packaging standards require that packaging materials are selected based on the nature of the drug. This ensures that the product remains stable from manufacturing to patient use.
For example, Vial Glass Bottle Boxes are commonly used for liquid medications because they offer reliable protection. For transportation, Corrugated Boxes are used to handle long-distance shipping while keeping the contents secure.
This level of protection is essential in the U.S., where clinical trials may involve multiple states and complex distribution systems.
Safety Features That Protect Patients
Safety is always the top priority in clinical trials. Packaging must clearly show if it has been opened or tampered with. This helps maintain trust and prevents the use of compromised products.
In some cases, additional safety measures are required. For example, Child Resistant Boxes are used when there is a risk of accidental exposure, especially in home-based trials.
The goal is to reduce risk as much as possible. Even small design features can make a big difference in protecting patients and ensuring that the trial runs smoothly.
The Importance of Organized and Scalable Packaging
Clinical trials often involve large numbers of participants across different locations. This makes organization and consistency very important.
Packaging must be easy to handle, store, and track. Solutions like Medicine Packaging Boxes help maintain uniformity and simplify distribution. In some cases, Hand Sanitizer Boxes are also included in trial kits to support hygiene protocols, especially in health-related studies.
A reliable custom Packaging Company plays a key role in managing these needs. They ensure that packaging is consistent across all batches and can scale as the trial expands.
What the Packaging of Investigational Drugs Should Ideally Deliver
At its core, the packaging of investigational drugs should ideally provide a balance of safety, clarity, and compliance. It should protect the drug, guide the user, and support accurate research outcomes.
Good packaging reduces errors, improves patient confidence, and helps researchers maintain full control over the study. It also ensures that the trial meets all regulatory expectations in the United States.
Final Thoughts
In today’s U.S. clinical environment, packaging is not just a technical requirement. It is a critical part of the healthcare system that supports both safety and innovation.
From following strict guidelines to ensuring product stability, every detail matters. Companies like Sire Printing that understand these requirements can provide real value by delivering packaging solutions that meet industry standards and support successful clinical trials.
When done correctly, packaging becomes more than just a container. It becomes a key factor in delivering safe and reliable healthcare solutions.
Frequently Asked Questions (FAQs)
What does the packaging of investigational drugs include?
The packaging of investigational drugs includes labeling, protective materials, storage instructions, tracking codes, and safety features that ensure proper handling during clinical trials.
Why is investigational drug packaging important in clinical trials?
It is important because it ensures patient safety, maintains drug stability, and supports accurate trial results by preventing mix-ups or contamination during testing.
What are investigational drug packaging requirements in the U.S.?
investigational drug packaging requirements in the U.S. include FDA-compliant labeling, tamper-evident design, proper storage conditions, and clear identification for trial use only.
How do clinical trial packaging guidelines improve accuracy?
clinical trial drug packaging guidelines help maintain consistency across all trial sites, reduce human error, and ensure that every participant receives the correct treatment.
What is pharmaceutical labeling compliance in the USA?
pharmaceutical labeling compliance USA refers to strict rules that require clear, accurate, and complete labeling of investigational drugs, including dosage, storage, and safety instructions.
What is investigational medicinal product packaging?
investigational medicinal product packaging refers to specialized packaging used to safely store, identify, and distribute drugs that are still under clinical testing and not yet approved for public use.
What are FDA clinical supply packaging standards?
FDA clinical supply packaging standards are regulations set by the U.S. Food and Drug Administration that ensure investigational drugs are safely packaged, labeled, and tracked throughout clinical trials.
Why are Custom Vinyl Stickers used in drug labeling?
Custom Vinyl Stickers are used because they are durable, easy to read, and remain intact under different storage conditions, helping maintain accurate and compliant labeling during trials.
What role does packaging play in clinical trial success?
Proper packaging ensures safety, prevents errors, maintains drug quality, and supports reliable data collection, which is essential for successful clinical trial outcomes.